|
Valence Electrons Chart for All Elements Atomic (Where you will get the HD images along with the explanation). If you want a Periodic table with Valence electrons, then visit Periodic table with Valence electrons labeled in it. But for most of the transition and inner transition elements, the valence electrons are the electrons present in the shells outside the noble gas core.For main group elements (i.e s-block and p-block elements), the valence electrons are the electrons present in the outermost orbit.The broom was discovered by Antoine Jérôme Balard in 1820. You can get valence electrons in an atomic arrangement of electrons by referring to the periodic table: atoms of group 1 have 1 valence electron. 
How many valence electrons f does it have?ħ valence electrons How many valence electrons does C have?įour valence electrons How many valence electrons does 2 have?Ģ answers. Oxygen belongs to group 6 and has 6 valence electrons. For example, carbon belongs to group 4 and has 4 valence electrons. The group number of an article can be found in the column of the periodic table. In the case of neutral atoms, the number of valence electrons is equal to the number of principal groups in the atom. (A semimetal shows the properties of the metal and the anonymous metal.) How do you find valence electrons? Although, like germanium and tellurium, it is actually a metal (more on this later in future articles), antimony is also known as a semimetal. Pure antimony is silver gray / white and brittle. The number of electrons in a neutral atom equals the number of protons. The number of protons in the atomic nucleus differs from the atomic number (Z). The number of protons, neutrons and electrons in an atom can be determined by a simple set of rules. The nucleus is made up of 51 protons (red) and 70 neutrons (yellow). Nuclear composition and electron configuration diagram of an antimony atom 121 (atomic number: 51), the most abundant isotope of this element. Which element has 51 protons and 70 neutrons? Nicolas Lémery, a French chemist, was the first to scientifically examine the testimonies and their connections. It is sometimes found freely in nature, but is generally extracted from the ores of stagnite (Sb2S3) and valentinite (Sb2O3). Who found the antimony?Īntimony has been known since ancient times. And the number of particles present in the nucleus is called the mass number (also called the atomic mass). Note that the atomic nucleus is made up of protons and neutrons. 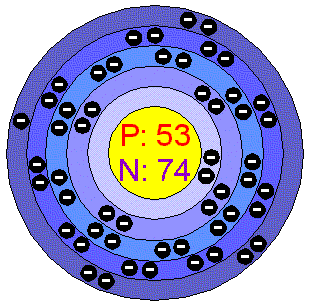
To find the number of neutrons in an atom, simply subtract the number of protons from the mass number. Cesium and Francium are the most reactive elements of this group. What is the second most reactive metal?Īlkali metals are softer than most other metals. 
Ubiquadium and Ubq are each the temporary name and symbol of the IUPAC until the element is recognized, verified, and a permanent name is established. Unbiquadium, also known as element 124 orecauranium, is the hypothetical chemical element with atomic number 124 and substitution symbol Ubq. Seven electronsHow many electrons are there in the ■■■■■■■ outer shell? fiveHow many electrons does xenon have in its outer shell? How many electrons does bromine have in the outer shell? How Many Valence Electrons Does Antimony Have How many electrons are there in the outer shell of antimony? | five electronsThat said, how many electron layers does antimony contain? data
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
